Epub ahead of print: Mandato et al. Safety of Endovascular Treatment of Chronic Cerebrospinal Venous Insufficiency: A Report of 240 Patients with Multiple Sclerosis. J Vasc Interv Radiol. 2011 Nov 14.
PURPOSE: To evaluate the safety of outpatient endovascular treatment in patients with multiple sclerosis (MS) and chronic cerebrospinal venous insufficiency (CCSVI).
MATERIALS AND METHODS: A retrospective analysis was performed to assess complications occurring within 30 days of endovascular treatment of CCSVI.
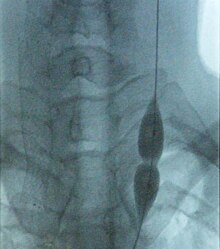
Fig: Balloon dilatation of stenosed jugular vein in an MS'er (source wikipedia)
RESULTS: After the procedure, all but three patients were discharged within 3 hours. Headache after the procedure was reported in 8.2% (21 of 257) of patients; headache persisted > 30 days in 1 patient. Neck pain was reported in 15.6% (40 of 257); 52.5% (21 of 40) of these patients underwent stent placement. Three patients experienced venous thrombosis requiring retreatment within 30 days. Sustained intraprocedural arrhythmias were observed in three patients, and two required hospital admission. One of these patients, who was being retreated for stent thrombosis, was hospitalized because of a stress-induced cardiomyopathy (abnormal heart function).
CONCLUSIONS: Endovascular treatment of CCSVI is a safe procedure; there is a 1.6% risk of major complications. Cardiac monitoring is essential to detect intraprocedural arrhythmias. Ultrasonography after the procedure is recommended to confirm venous patency and to identify patients experiencing acute venous thrombosis.
"The question is how safe is safe enough? An impossible call as we have no data on whether or not this treatment results in a real benefit or is simply due to the placebo effect. We have to wait for sham treatment trials to answer the latter."
"To reiterate I don't recommend investigation or treatment for CCSVI unless it is part of a clinical trial that has been vetted and approved by an ethics committee, you give informed consent, there is insurance in place to protect you from any mishaps that may occur and you don't have to pay to participate in the study."