Mitochondria are essential to neuronal viability and function due to
their roles in ATP production, intracellular calcium regulation, and
activation of apoptotic pathways. Accordingly, mitochondrial dysfunction
has been indicated in a wide variety of neurodegenerative diseases,
including Alzheimer's disease (AD), Huntington's disease, amyotrophic
lateral sclerosis, stroke, and multiple sclerosis
(MS).
Recent evidence points to the permeability transition pore (PTP)
as a key player in mitochondrial dysfunction in these diseases, in which
pathologic opening leads to mitochondrial swelling, rupture (bursting), release of
cytochrome c (an oxygen binding molecule that can transfer electrons in the electron transport chain), and neuronal death. Reactive oxygen species (ROS=radicals), which
are inducers of PTP opening, have been prominently implicated in the
progression of many of these neurodegenerative diseases. In this
context, inactivation of a mitochondria-targeted redox (reduction and oxidation) enzyme p66ShcA
(p66) has been recently shown to prevent the neuronal cell death leading
to axonal severing in the mouse model of MS, experimental autoimmune
encephalomyelitis (EAE).
To further characterize the response of neurons
lacking p66, we assessed their reaction to treatment with stressors
implicated in neurodegenerative pathways. Specifically, p66-knockout
(p66-KO) and wild-type (WT) neurons were treated with hydrogen peroxide and nitric oxide (both reactive oxygen species), and assessed for cell viability and
changes in mitochondrial properties, including morphology and ROS
production. The results showed that p66-KO neurons had greater survival
following treatment with each stressor and generated less ROS when
compared to WT neurons.
Overall, these findings highlight the importance of developing
mitochondria-targeted therapeutics for neurodegenerative disorders, and
emphasize p66, mitochondrial ROS, and the PTP as key targets for
maintaining mitochondrial and neuronal integrity.
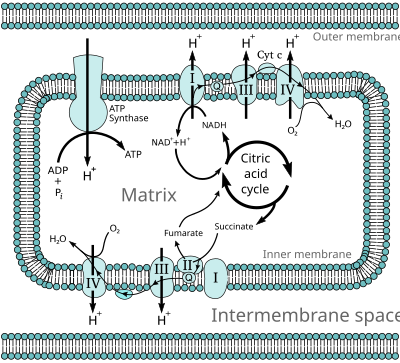
The electron transport chain in the mitochondrion is the site of oxidative phosphorylation in eukaryotes. The NADH and succinate generated in the citric acid cycle are oxidized, providing energy to power ATP synthase.
Mitochondria are structures that convert the chemical energy from food and oxygen to a form of energy (adenosine triphosphate (ATP)) that cells can use. If cells do not get enough energy they may die (apoptosis = cell suicide). They are central to nerve function and mitochondrial deficits have been found in demyelinated nerves in MS and this may leave them susceptible to nerve loss. This study looks at an enzyme that is involved in the regulation of free radicals and may protect from nerve death. This may be a target for control of nerve damage in progression.
This is an area of active research including Team G.