CISers have evidence of early brain shrinkage. #MSBlog #MSResearch
Epub: Pérez-Miralles et al. Clinical impact of early brain atrophy in clinically isolated syndromes. Mult Scler. 2013 May 7.
BACKGROUND: The impact of global and tissue-specific brain atrophy on conversion to MS after a clinically isolated syndrome (CIS) is not fully gauged.
OBJECTIVES: These investigators' aimed to determine the magnitude and clinical relevance of brain volume dynamics in the first year after a CIS.
METHODS: They assessed 176 CISers within 3 months of onset, clinically and by conventional magnetic resonance imaging (MRI) scans, at baseline and 1 year after clinical onset. They determined the percentage of brain volume change (PBVC) and the brain parenchymal (BPF), grey matter (GMF) and white matter (WMF) fractions.
PBVC (percentage of brain volume change) = proportional change in whole brain without the cerebrospinal fluid spaces relative to baseline
BPF (brain parenchymal) = the actual volume of the brain without the cerebrospinal fluid spaces
GMF (grey matter fraction) = includes the cortex (surface of brain) and deep gray matter structures
WMF (white matter fraction) = the areas of the brain that are not defined as grey matter
RESULTS: The mean follow-up time was 53 months (SD = 16.8): 76 CISers (43%) experienced a second attack, 32 (18%) fulfilled MRI-only 2005 McDonald criteria and 68 (39%) remained as CIS. Statistically significant decreases in the volume measures tested were observed in CISers with a second attack, for BPF and PBVC; in both MS groups for GMF; whereas in all groups, the WMF was unchanged. CISers with a second attack had larger PBVC decreases (- 0.65% versus + 0.059%; p < 0.001). PBVC decreases below - 0.817% independently predicted shorter times to a second attack.
CONCLUSIONS: Global brain and grey matter volume loss occurred within the first year after a CIS; brain volume loss predicted conversion to MS.
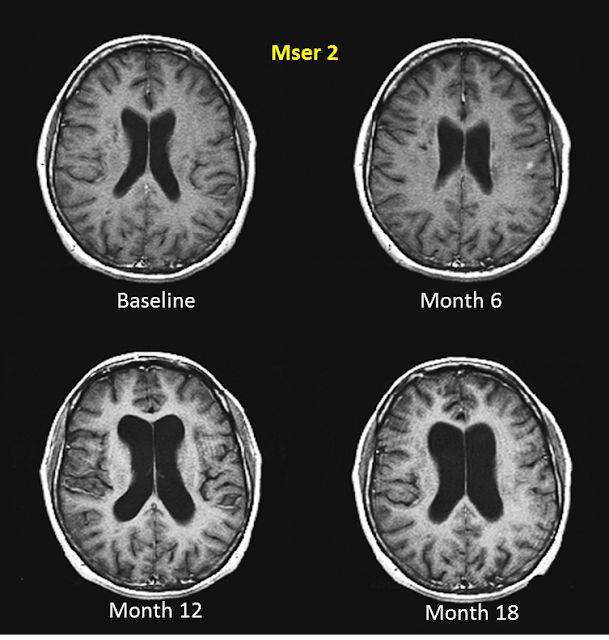 |
| Brain atrophy over 18 months in an RRMSer |
"This study confirms the results of previous studies in this area. It is clear that at first presentation CISers have evidence of brain damage as measured by brain atrophy. The degree of brain atrophy is able to predict who will have second attack; this is not surprising and simply tells us that CISers with the most active disease are the ones that are more likely, statistically, to have a second attack."
"What is interesting is that regardless of whether or not these CISers had a second attack, or not, they went onto develop progressive gray matter atrophy over the next few years. Why is this important? This is important for two reasons. Firstly, gray matter atrophy, in particular the cortex or surface of the brain, is linked to cognitive impairment. Cognitive impairment is the reasons why CISers and MSers have problem with studying and work. Secondly, grey matter atrophy is important as many neurologists still don't treat CISers with DMTS; either they don't think it is necessary or the local/national guidelines don't allow it."
"We now have drugs that impact on grey matter atrophy. Who sets these prescribing guidelines that prevents neurologists prescribing DMTs for CIS? In the UK they are set by the National Institute for Health and Care Excellence (NICE) with the backing of outdated Association of British Neurologists guidelines. The implications is that CISers are being denied early treatment. At the American Academy of Neurology meeting in San Diego there was a poster showing that delaying DMTs in CISers until the second attack had a significant cost associated with it; CISers in whom treatment was delayed did worse than those started on treatment immediately. It is time neurologists started lobbying payer and organisations to adopt the early aggressive treatment strategy."
"What is really depressing are the results of the Cladribine in CIS study, or ORACLE Study, that were presented at the AAN. These results are the best we have seen to date in CIS and clearly make a real difference. Some of the CISers treated with cladribine may never have a second attack; I suspect we will never get to hear the information. The real tragedy is that the company behind cladribine has decided to pull the plug on the drug. Why? Business, the patent life on the drug is too short to justify its further development. Can we repurpose Cladribine and salvage it for MSers? Possibly but it is going to take a massive effort and commitment from the MS community."