Biological treatment with inhibitors of the pro-inflammatory cytokine TNF-alpha has dramatically improved the disease course of several chronic rheumatologic conditions. Adverse events (AEs) are primarily infections and hypersensitivity reactions. Demyelinizing neurological symptoms resembling multiple sclerosis (MS) have been described as a rare AE. During about 10-year use of anti TNF-alpha, the Danish Medicines Agency has recorded eight cases of MS like AEs. The objective of this study was to estimate the incidence of demyelinizing AEs both in the central and peripheral nervous system after treatment with anti TNF-alpha in a cohort of patients from a large rheumatologic outpatient clinic in Copenhagen. In a 4-year period from January 2008 to December 2011, approximately 550 patients annually were undergoing treatment with anti TNF-alpha inhibitors in our department. We collected data on all patients who developed neurological symptoms during this time period. We found six patients with signs of demyelinizing neurological disorders: four resembling MS, one MS-like condition, and one multifocal motor neuropathy. During a relatively short time period, we found a remarkably high number of neurological demyelinizing AEs probably linked to anti TNF-alpha treatment. The AEs were not associated with a single anti TNF-alpha agent and were thus presumably a class effect. The data presented suggest that neurological AEs may be underreported. We advocate that physicians handling patients during treatment with TNF inhibitors are aware of this potentially serious AE and report these events to the proper medical authorities
We have been talking about the risks of extra autoimmunities that occur following the treatment with Alemtuzumab. At present about 20-30% of MSers develop secondary autoimmunities after antibody treatment. The FDA considers this too much of a risk.
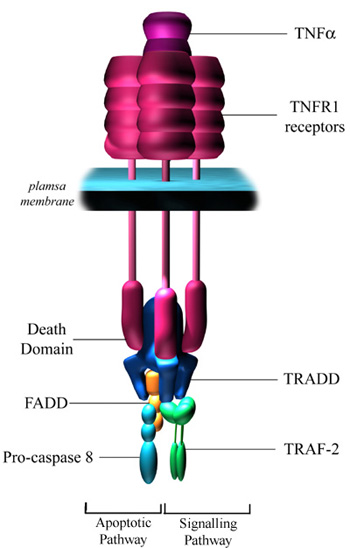
However it is not just MSers that can get additional autoimmunity following treatment. Tumour necrosis factor alpha inhibitors have been a wondrous treatment for rheumatoid arthritis, however this comes at a cost too. In this case, one of the problems in the treatment of Arthritis is the risk of developing MS. This occurs in about 1 in 100. People can also get Lupus an autoimmune disease against DNA. This effect has been seen with other inhibitors of TNF such as some Phosphodiesterase Type IV inhibitors