Is Prof G a laggard? Is he being too conservative by not embracing HSCT as an MS treatment? #ClinicSpeak #MSReserch #MSBlog
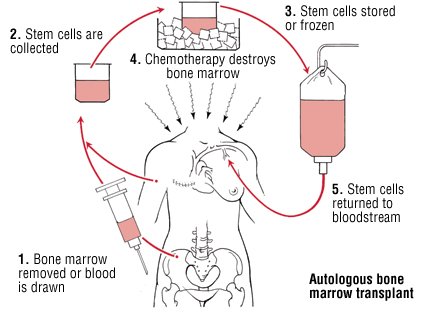
"It is clear from reading comments on several recent posts, including yesterday's post, that a large number of you would prefer the option of non-myeloablative, or ablative, hematopoietic stem transplantation (HSCT) to alemtuzumab treatment. Is that correct? HSCT looks to have the same or higher efficacy than alemtuzumab treatment and seems to come with a lower risk of secondary autoimmunity. HSCT is clearly more expensive initially, but may prove more cost-effective over time, particularly if a larger proportion of MSers treated with HSCT go into long-term remission compared to alemtuzumab. The problem we have as an MS treatment centre is that HSCT is not a licensed treatment for MS and hence our hospital, or Trust (Barts Health), would have to cover the costs of the treatment. In comparison, the costs of alemtuzumab, a licensed MS treatment, is covered by NHS England and not our own hospital. Under the current financial constraints in the NHS our managers would, almost certainly, prevent us offering HSCT to MSers as part of routine care. However, if we did this as part of a clinical trial and got the NIHR (National Institute for Health Research) to fund the treatment we would be able to offer this as a treatment for MS. However, this would have to be under a research protocol hence there is no guarantee that you would be randomised to the HSCT arm. Do you think the NIHR should a trial of this kind? It would have to be a pragmatic non-inferiority, or safety, study; trying to show that HSCT is superior to Alemtuzumab using our current outcomes would be very difficult and prohibitively large."
"Back in 1998, when I had moved to the Royal Free Hospital, for a short period of time, I did due diligence on bone marrow transplantation in MS and other autoimmune conditions. At the time there was a very active group at the RFH doing BMT to treat a host of other autoimmune diseases, in particular systemic sclerosis and SLE. When I saw that the mortality from having a BMT in autoimmunity was between 2-5% I decided back then that the risks outweighed the benefits in relation to MS. However, since then the risks associated with BMT and HSCT have plummeted and the mortality in good units is well below 0.5% from the procedure (less than 1 in 200). The question is are these risks worth it? I suspect yes. On my recent visit to Canada I discovered that several Canadian centres are now offering HSCT as part of their routine service to MSers with highly-active MS. On the spectrum of neurological conservatism the Canadian neurologists are very similar to UK neurologists; I would therefore expect that some UK neurologists and MSers with opt for this treatment option if it was available. Therefore it may be time for neurologists in the UK to join the Canadians, Americans and other countries (and the handful of centres in the UK) and start offering HSCT to our MSers with highly-active MS, who prefer the risks associated with HSCT over the risks of alemtuzumab treatment."
"I find it very interesting that a large number of you have criticised me for being so gung-ho over my attitude to using highly-effective treatments early in the course of MS and yet others of you are saying that I am too conservative, and a laggard (non-adopter), over my attitude to HSCT. The reason I have been slow to adopt HSCT and remain conservative is because anti-CD20 treatment may change things. Anti-CD20 as a class of therapies may prove to be so good at controlling MS disease activity, with a much better safety profile compared to alemtuzumab, natalizumab and HSCT that it would be hard to justify more risky therapies outside of the small group of MSers who breakthrough on an anti-CD20 therapy. Ocrelizumab, and the other anti-CD20 and anti-CD19 monoclonals (also a B cell therapy), may change the treatment paradigm and the risk-benefit of highly-effective treatments to such an extent that some of these arguments above would be academic. I think the arrival of ocrelizumab as a treatment for relapsing-MS, and possibly PPMS, will be the real game-changer in relation to risk:benefit. I sincerely hope Roche have a heart and don't price the drug too high taking it out of the reach of most MSers. What I am most interested in knowing is how ocrelizumab and the other anti-CD20 therapies work? Therein lies the clue to what causes MS."
CoI: multiple