INTRODUCTION:Multiple sclerosis (MS) is an immune-mediated disease. Over the last decades therapeutic options have broadened tremendously. Nevertheless, various therapeutic agents, e.g., rituximab, are currently used in the treatment of MS off label. Disease or health registries are useful methods to collect information about off-label treatments. The German registry for autoimmune disease (GRAID) is a multicenter, retrospective, non-interventional database of patients with various autoimmune diseases.
AIM/METHODS: The aim of this observational analysis is to present safety data of rituximab in the treatment of MS and neuromyelitis optica (NMO) in a real life clinical setting based on the available registry data.
RESULTS: Data were collected nationwide in patients who received rituximab. 56 patients were treated with rituximab for MS or NMO. Average observation period was 9.6 months (SD 7.6, ranging from 6 to 29.7 months). Interval between treatments cycles differed tremendously (ranging from 0 to 21 months, median 10 months). Number of infusions ranged from 1 up to more than 8. The analysis provides experience on almost 50 patient years. Infusion related reactions were most common and reported in four patients; infections were seen in three patients (two of them were hospitalized for urinary tract infection and urosepsis). All patients recovered from infection. Full treatment response was attested in a quarter of the patients; two thirds benefited partially from treatment.
DISCUSSION: Safety data of almost 50 patient years of treatment with rituximab show that rituximab is tolerated well in MS/NMO patients. Infections and infusion reactions are the most common adverse events. Our data may help the individual physician to balance efficacy of rituximab against the risk. • Data on rituximab in MS and NMO are provided for almost 50 patientyears • Rituximab was tolerated well • No unexpected side effects were seen • Almost 80 % of the patients benefited at least partially from treatment.
AIM/METHODS: The aim of this observational analysis is to present safety data of rituximab in the treatment of MS and neuromyelitis optica (NMO) in a real life clinical setting based on the available registry data.
RESULTS: Data were collected nationwide in patients who received rituximab. 56 patients were treated with rituximab for MS or NMO. Average observation period was 9.6 months (SD 7.6, ranging from 6 to 29.7 months). Interval between treatments cycles differed tremendously (ranging from 0 to 21 months, median 10 months). Number of infusions ranged from 1 up to more than 8. The analysis provides experience on almost 50 patient years. Infusion related reactions were most common and reported in four patients; infections were seen in three patients (two of them were hospitalized for urinary tract infection and urosepsis). All patients recovered from infection. Full treatment response was attested in a quarter of the patients; two thirds benefited partially from treatment.
DISCUSSION: Safety data of almost 50 patient years of treatment with rituximab show that rituximab is tolerated well in MS/NMO patients. Infections and infusion reactions are the most common adverse events. Our data may help the individual physician to balance efficacy of rituximab against the risk. • Data on rituximab in MS and NMO are provided for almost 50 patientyears • Rituximab was tolerated well • No unexpected side effects were seen • Almost 80 % of the patients benefited at least partially from treatment.
So this is more data in a prelude to ocrelizumab arriving and maybe putting an end to rituximab use for MS, if/when ocrelizumab gets a licence. There are over 1,000 people treated with this in Sweden.
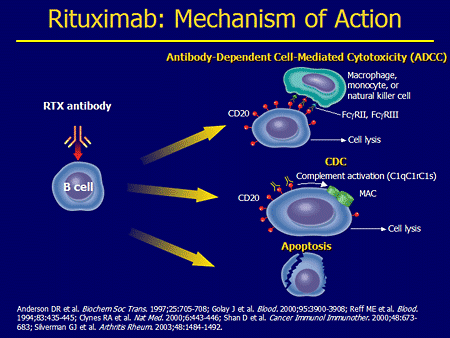
One of the problems with rituximab is that the antibody is chimeric and so has mouse bits in it. Apparently about 30% of people can make an anti-globulin response to this as the antibody mechanism does not protect itself, whereas a anti-CD4 antibody response can stop anti-globulin responses occurring. Ocrelizumab is humanised and apparently the anti globulin response drops dramatically, whiich makes it useful for long term use. The ofatuzumumab on the way too is fully human and so this reaction should drop further. The anti-globulin response will contribute to the drug not working as well as it could.
It is cealr that relapsing and active progressive MSers can benefit from this type of treatment, and so it is wrong that criteria for stopping DMT is onset of progression.