We are also very aware that there is a growing voice of resistance to the use of animals and amongst the most vocal are the clinical fraterinity...who chunter "animal studies are a waste of time, they haven't ever found anything useful". I have been listening to this for the past decade or more but sadly people in power are beginning to listen and it is becoming increasing difficult to get animal studies supported.
I personally get fed up with these witterings because it is clear to me that the reverse can also be said and neurologists can contribute to studies not translating, such as through inapproapriate trial design and implementation. However, is it taboo to say such a thing. Whilst it took some time to put these concerns into print in, contrast to the animal bashing that some (arrogant) people have been happy to dish out over the years.
However, basic scientists are contributing to their own demise as there are a number of animal studies that have poor translatability into human use and we should not be adverse to point out problems because unless the basic scientitsts change their ways, they are fueling the knockers of animal experiments.
We are called all sorts of things for commenting on the fragility of animal studies and get stick from people because of this. However we want to be constructive and help people better interpret their own data and want to encourage the funders to support good quality animal studies. If you are a scientist and think it doesn't matter...fine but you are doing your bit to hasten your own demise
One aspect of concern is the experimental design and the way the experiments are reported, such that you can see if bias was minimised. This is central to a clinical trial but seldom is sufficient information given so that you know what was done in animal experiements
We know that experimental bias is central to over exaggerated animal studies, which won't be reproduced in another lab, let alone in a human.
The ARRIVE guidelines are a set of 20 reporting issues that aim to improve the transparency of the experiments. CLICK HERE TO READ THEM There has been resistence to adopting these. I think some of this is because they are not "made in America" (and this is not the way we do it) and for some aspects they ask people to do things that are not current practise.
In MS animals studies the mantra seems to be let's do 3 experiments with n=5 in a group. Rather than do a sample size calculation to determine how many animals should be used to see a desired effect. N=5 may be hopelessly underpowered
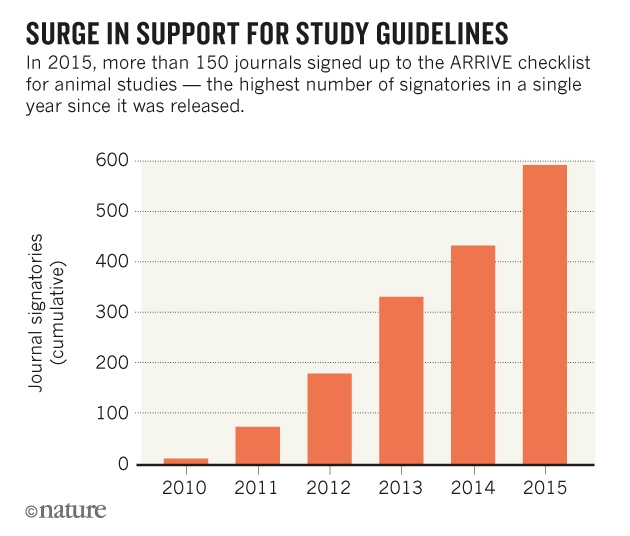
However, it is clear that it is slowly catching on and more and more journals are signing up to this. Nature jounrals were among the worse offenders of non-tranparent methodology now they ARRIVE standard reporting and want their own check list filled out before you can submit.
It is clear that more an more journal are endorising this and with time papers will change otherwise they won't get published. If papers get turned down then people will start to change. Ten years ago there were few papers that mentioned ethical review of their work. Now it is virtually all that report it. It is simply down to a matter of enforement.
However it is clear that there is increasing interest in this (CLICK)
If journals are going to endorse the guidelines they need to enforce them too, because otherwise it is pointless lip service.
The number of papers I see where it states "We did the experiments according to the ARRIVE guidelines" This is mushroom food. because although by reporting the experimental design it may improve it, the point is reporting what you do, not dictating what you do. So you don't blind experiments...no problem but it means that the results may be more likely to be biased and so not reproducible.
Pharma support the ARRIVE guidelines, charities support the ARRIVE guidelines because it supports better laboratory practice. Some labs may not like it...one has to then ask, is it because they are producing unreproducible guff that fuels those descenting voices?
So when you read papers....think! Do the animal studies report aspects relevant to the ARRIVE guidelines they may help you decide whether to believe the findings.
Is 600g of salt a day relevant human biology? or should you be prepared to take it with a pinch?
Is 600g of salt a day relevant human biology? or should you be prepared to take it with a pinch?
If the concern is whether the animal experiments are going to translate into human benefit, the simple way to get enforcement is not through the journals but through the patenting system.
Most animal studies are about finding mechanisms of disease and not about showing the value of treatments.
The Governments want transparent animal experiments with impact, if this was a reporting standard required for a patent, and it wasn't done to that standard then the work could be used to justify an approach for human studies and it would lack the impact that the Government want. If you cannot file a patent unless ARRIVE guidleline aspects are reported, Universities will soon encourage their workers to do this.
If only the US or European patent offices would do this so you only have to lobby two places and get them to agree and have to lobby two thousand journals. Without patents to protect the US and European markets any work not doing this would be commercially worthless. This should make governmenetns happy, pharma would be happy because they do their experiments to good standards and so protocols would be easy to report, academia would have to toe the line.
Most animal studies are about finding mechanisms of disease and not about showing the value of treatments.
The Governments want transparent animal experiments with impact, if this was a reporting standard required for a patent, and it wasn't done to that standard then the work could be used to justify an approach for human studies and it would lack the impact that the Government want. If you cannot file a patent unless ARRIVE guidleline aspects are reported, Universities will soon encourage their workers to do this.
If only the US or European patent offices would do this so you only have to lobby two places and get them to agree and have to lobby two thousand journals. Without patents to protect the US and European markets any work not doing this would be commercially worthless. This should make governmenetns happy, pharma would be happy because they do their experiments to good standards and so protocols would be easy to report, academia would have to toe the line.